FMC Corporation (NYSE:FMC) today reported first quarter 2026 revenue of $759 million, down 4 percent versus first quarter 2025. First quarter 2026 revenue, excluding India, was $762 million, down 4 percent versus first quarter 2025, which included India. On a GAAP basis, the company reported a loss of $2.25 per diluted share in the first quarter, a decrease of $2.13 versus first quarter 2025. First quarter adjusted loss per diluted share of $0.23 was down 41 cents versus first quarter 2025.

First quarter sales of $762 million, excluding India, were above the midpoint of guidance and 4 percent lower than the prior year. The removal of India represented a 5 percent sales headwind. Price declined 6 percent, in line with expectations, driven by lower pricing to diamide partners, pricing actions on branded Rynaxypyr® products and a competitive market for legacy core products – particularly in Latin America. Foreign currency was a tailwind of 5 percent. Volume improved 2 percent, driven by strong growth in EMEA and North America. New active ingredient sales doubled year-over-year. Plant Health grew 6 percent.
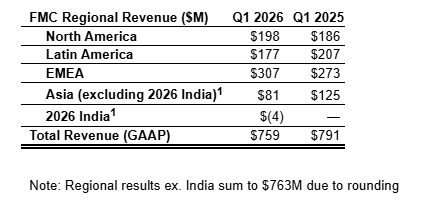
GAAP net loss in the first quarter declined $266 million primarily due to tax charges related to an increase in valuation allowances. Lower sales, higher restructuring costs and higher interest expense also contributed to the loss during the first quarter. FMC first quarter Adjusted EBITDA was $72 million, a decrease of 40 percent from the prior-year period, driven by lower pricing and unfavorable costs. The cost increase was driven by tariffs as well as unfavorable raw material costs.
On a GAAP basis, cash from operations was negative $601 million, a decline of $56 million versus 2025, primarily driven by lower Adjusted EBITDA. Free cash flow was negative $628 million, a decline of $32 million versus Q1 2025 primarily due to lower cash from operations, partially offset by lower capital expenditures.
Strategy Update
FMC is making strong progress on its 2026 operational priorities, which are strengthening the balance sheet through targeted debt reduction of approximately $1 billion, improving the competitiveness of its core portfolio, managing the post-patent transition for Rynaxypyr® active, and driving growth of new active ingredients including Isoflex® active, fluindapyr and Dodhylex® active. In parallel, the Board-authorized evaluation of strategic alternatives announced in February 2026 is progressing, and multiple options are being evaluated. There can be no assurance that the process will result in any transaction. The company does not intend to comment further at this time, except as it may do so in the ordinary course in connection with its upcoming earnings call, or if it determines that further disclosure is appropriate or necessary.
Full Year Outlook1
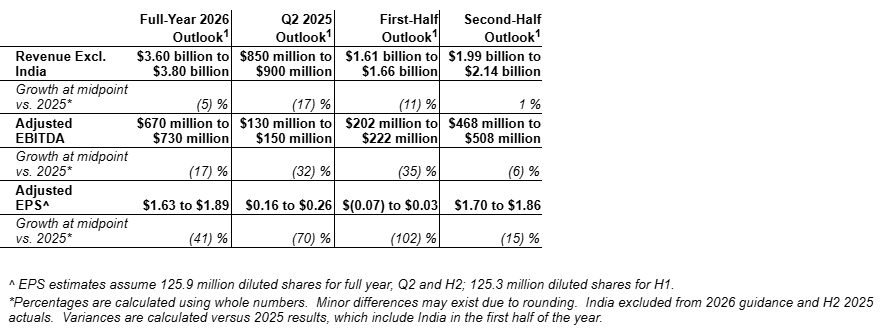
The company reaffirms its full-year 2026 revenue, Adjusted EBITDA, Adjusted EPS and free cash flow guidance ranges. Full year 2026 revenue guidance1 is $3.60 billion to $3.80 billion, a decline of 5 percent at the midpoint versus prior year1. Price is expected to be lower by mid-single digits mainly due to Rynaxypyr® active, which is consistent with the company’s post-patent strategy. Excluding India, volume is expected to be up modestly as increases in branded Rynaxypyr® active and new active ingredients are largely offset by reduced diamide partner orders and declines in the legacy core portfolio. India represents a 2 percent headwind1. FX is expected to be neutral. Sales of new active ingredients are expected to be between $300 million and $400 million, representing growth of over 75 percent at the midpoint versus prior year.
Adjusted EBITDA is expected to be $670 million to $730 million, a decline of 17 percent versus prior year as lower price and an FX headwind are partially offset by volume growth and favorable costs. EPS is expected to be $1.63 to $1.89, a decrease of 41 percent versus prior year, primarily due to lower Adjusted EBITDA and, to a lesser extent, increased interest expense. Free cash flow is expected to be negative $65 million to $65 million.
Second Quarter and H2 Outlook1
Second quarter revenue is expected to be in the range of $850 million to $900 million, a decline of 17 percent at the midpoint compared to second quarter 2025, primarily due to lower volume to diamide partners and the removal of India. The India inclusion in prior year represents a 5 percent headwind. Price is expected to decline mid-single digits due to competitive pressure and planned pricing actions for Rynaxypyr® in line with the post-patent strategy. FX is expected to be a low-single digit tailwind. Adjusted EBITDA is forecasted to be in the range of $130 million to $150 million, a decline of 32 percent versus the prior year as lower sales are partially offset by favorable costs. FMC expects Adjusted EPS to be in the range of $0.16 to $0.26 in the second quarter, which represents a 70 percent decrease at the midpoint versus second quarter 2025, due to lower Adjusted EBITDA as well as higher interest expense to a lesser degree.
The midpoint of first-half guidance implies a second-half sales increase of 1 percent versus prior year. Price is expected to be a mid-single digit headwind, driven by competitive market conditions for core portfolio products and pricing actions to support the branded Rynaxypyr® active strategy. Lower price and a minor FX headwind are expected to be more than offset by volume growth, driven primarily by increased sales of products with new active ingredients.
Second-half Adjusted EBITDA is expected to decrease 6 percent as lower price and a minor FX headwind are partially offset by higher volume and favorable costs. Second-half Adjusted EPS is expected to decline 15 percent compared to second half 2025, due to lower Adjusted EBITDA, higher tax, and higher interest expense.
Supplemental Information
The company will post supplemental information on the web at https://investors.fmc.com, including its webcast slides for tomorrow’s earnings call, definitions of non-GAAP terms and reconciliations of non-GAAP figures to the nearest available GAAP term.
Always read and follow all label directions, restrictions and precautions for use. Products listed here may not be registered for sale or use in all states, countries or jurisdictions. FMC and the FMC logo are trademarks of FMC Corporation or an affiliate.


















